Arginine, Ornithine and Citrulline in Plasma
April 6, 2026 2026-04-07 11:04
Arginine, Ornithine and Citrulline in Plasma
For research/analytical guidance only. Not for diagnosis or clinical decision-making.
Until recently, monitoring urea-cycle amino-acid metabolites such as arginine, ornithine, and citrulline in plasma samples from preclinical and clinical studies largely relied on LC-MS- and HPLC-based methods. While highly powerful, these workflows are often outsourced because they require specialized instrumentation, dedicated expertise, and relatively complex analytical setups.
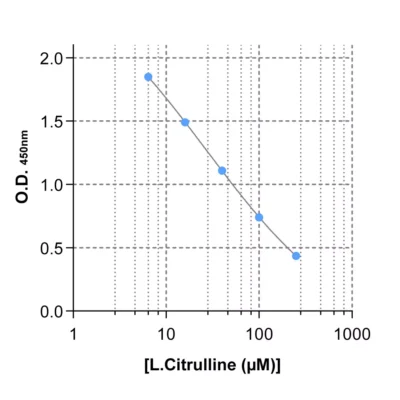
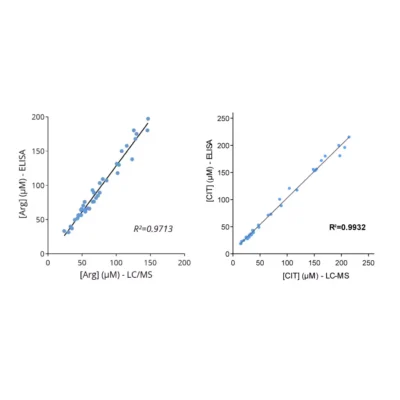
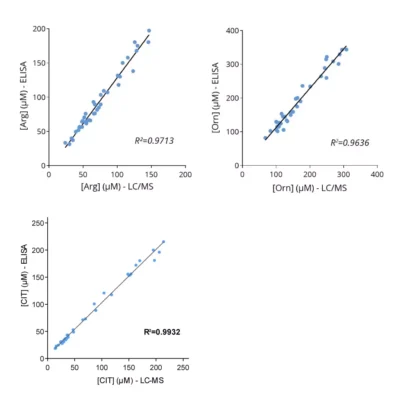
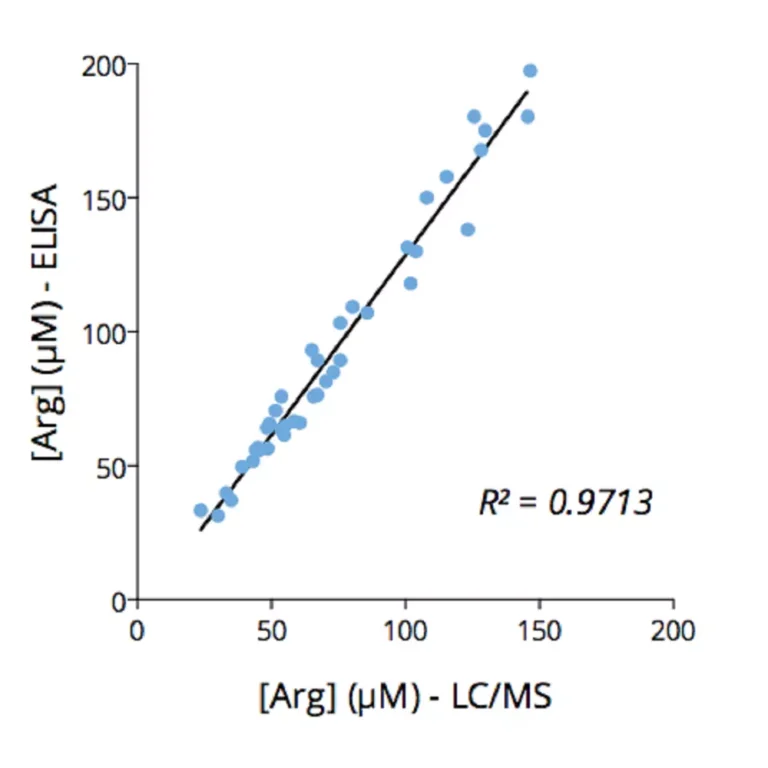
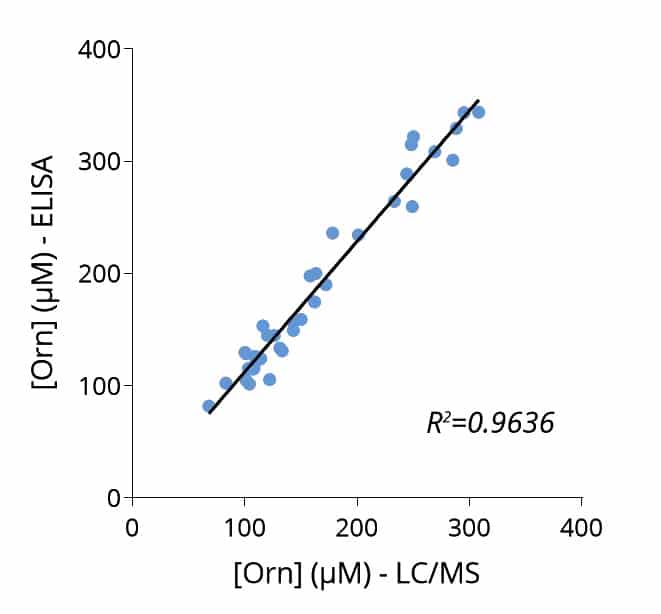
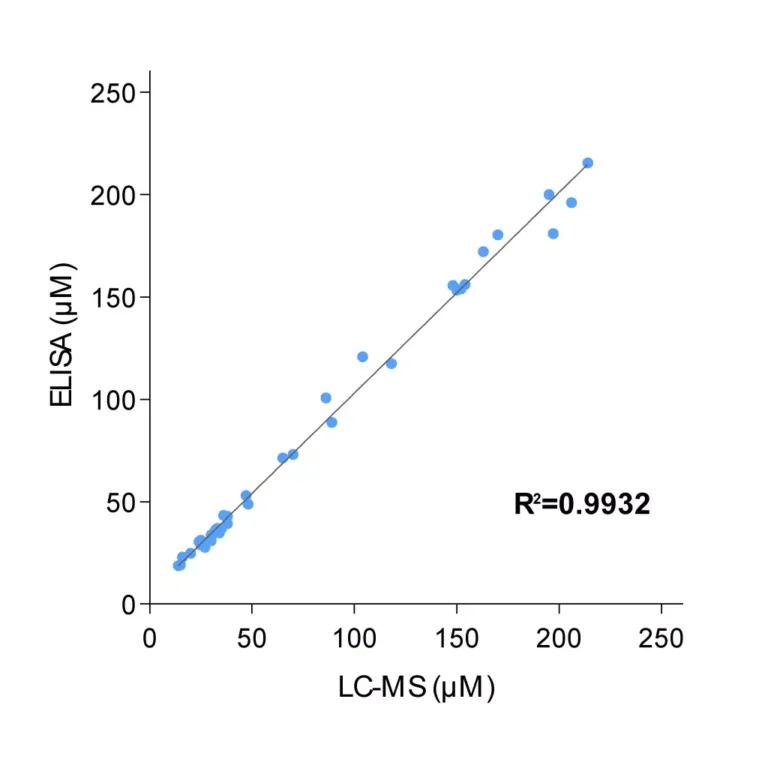
To enable simpler and higher-throughput quantification across animal and human samples, Immusmol developed dedicated ELISA kits for the measurement of arginine, ornithine, and citrulline in plasma. These assays are cross-species compatible, require only low sample volumes, and are therefore well suited to rodent studies. They were also systematically cross-validated against LC-MS in human plasma samples to ensure accurate and robust results.
Why measure arginine, ornithine and citrulline together in plasma?
Arginine, ornithine, and citrulline are closely linked metabolites within the arginine–urea cycle and nitric oxide pathways. Arginine is converted by nitric oxide synthase into citrulline and nitric oxide, and by arginase into ornithine and urea. Because these pathways compete for the same substrate, measuring the three metabolites together can provide a more informative view of pathway activity than single-analyte quantification alone.
In plasma, this combined approach is particularly useful because it enables calculation of biologically relevant ratios that help interpret arginine bioavailability and pathway balance. Immusmol’s ELISA workflows therefore enable both the quantification of arginine, citrulline, and ornithine in plasma and the calculation of key arginine-related ratios:
Applications in oncology
Arginine metabolism lies at the intersection of tumor-cell metabolism, myeloid-driven immune suppression, and therapeutic vulnerability. In the tumor microenvironment, arginase-expressing myeloid cells can deplete extracellular arginine and generate ornithine, a mechanism known to suppress T-cell responses while also feeding downstream pathways linked to tumor growth, including polyamine synthesis.
In this context, plasma arginine can be used as a systemic readout of arginine availability, while ornithine provides complementary information on arginase-driven catabolism. Measuring both metabolites together, and especially calculating the arginine/ornithine ratio, can therefore help interpret whether reduced arginine bioavailability may be linked to increased arginase pathway activity. This makes the arginine–ornithine axis particularly relevant in translational oncology and cancer immunology studies.
A second major application is the pharmacodynamic monitoring of arginine-targeting therapies. In clinical studies of pegylated arginine deiminase, circulating arginine has served as a direct pharmacodynamic marker, with treatment-associated decreases in plasma arginine and increases in citrulline documenting target engagement and systemic metabolic effect. This has been demonstrated in malignant pleural mesothelioma and explored in early-phase studies in non-squamous NSCLC, high-grade glioma, and uveal melanoma.
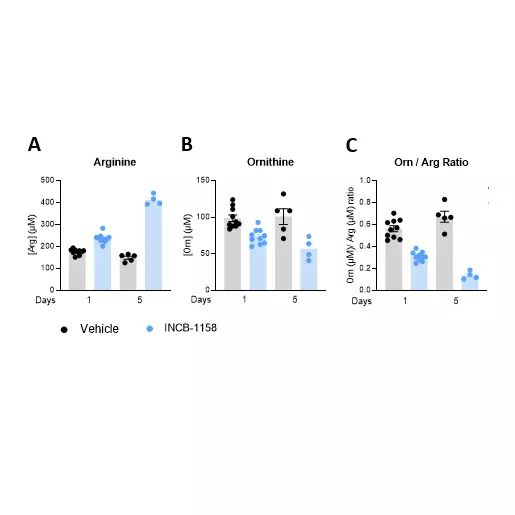
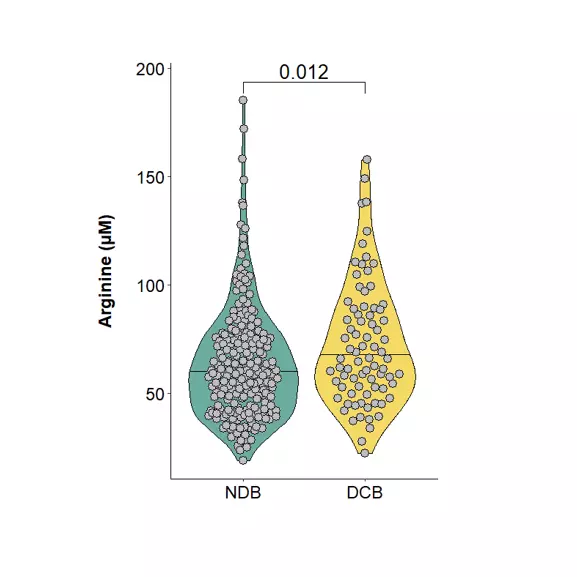
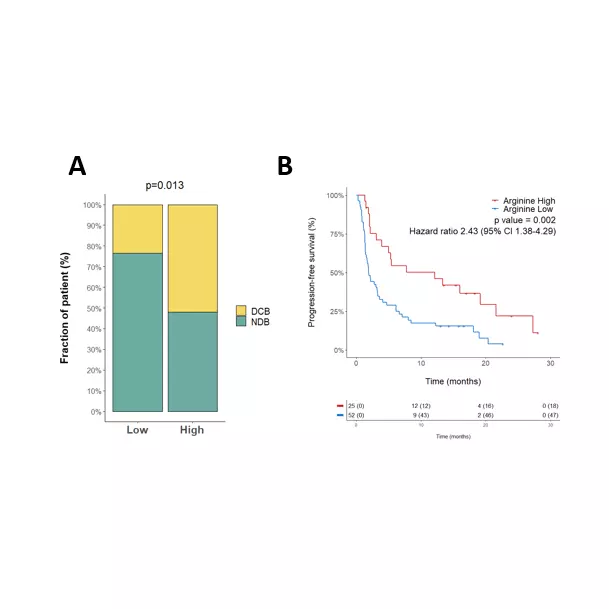
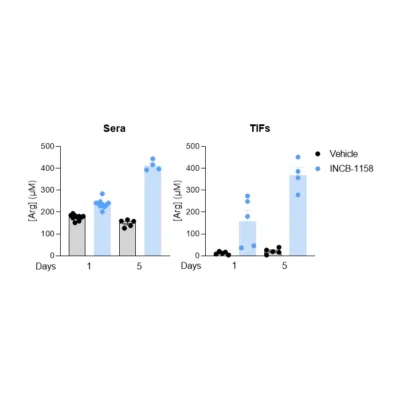
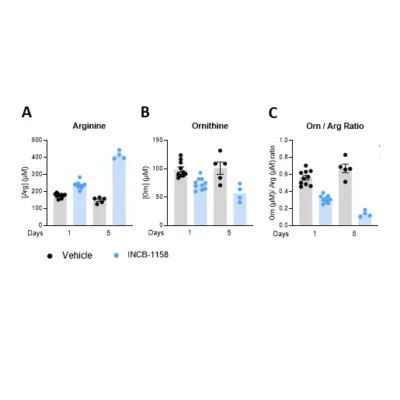
Our team also reported in a 2022 Annals of Oncology paper that baseline plasma arginine measurement before cancer immunotherapy is clinically relevant (see figures above). We showed that low baseline circulating L-arginine in advanced cancer was associated with poorer outcomes after immune checkpoint inhibitor treatment, supporting plasma arginine as a promising predictive biomarker of ICI response. Beyond ICIs, other T-cell-dependent therapies, including bispecific T-cell engagers (BiTEs), may also benefit from assessment of arginine availability.
Taken together, plasma monitoring of arginine-pathway metabolites in oncology can support several applications:
characterizing systemic arginine availability in cancer patients
assessing arginase-driven immune suppression
supporting interpretation of ornithine-linked metabolic rewiring
monitoring pharmacodynamic effects of arginine-targeting therapies
exploring baseline arginine as a predictive or stratification biomarker before immunotherapy
Applications in immunometabolism and inflammation
Plasma arginine measurement is particularly relevant in inflammation because circulating arginine frequently decreases during systemic inflammatory states, and this decrease has been repeatedly linked to disease severity. In sepsis, a systematic review and meta-analysis found that plasma arginine concentrations were significantly lower in patients than in controls, with an average reduction of about 34 µmol/L. The same review concluded that hypoargininemia is a consistent feature of sepsis.
This pattern is also observed in critically ill children. In a 2007 study, plasma arginine concentrations were markedly lower in children with sepsis and trauma than in children with viral illness, and both arginine and citrulline showed strong inverse correlations with CRP, linking these metabolites directly to inflammatory burden. During recovery, both metabolites increased again as inflammation subsided.
These observations make plasma arginine useful as a systemic biomarker of inflammatory stress and arginine deficiency, especially in settings such as sepsis, endotoxemia, and ICU research. Citrulline can add value in the same context, because low plasma citrulline has also been described during acute inflammatory states and may reflect broader impairment of the arginine–citrulline–NO axis.
Beyond acute inflammation, plasma arginine-pathway markers have also shown relevance in chronic inflammatory disease. In rheumatoid arthritis, one study reported increased plasma ornithine, elevated plasma arginase activity, and reduced global arginine bioavailability, supporting the idea that chronic inflammation can shift circulating arginine metabolism toward arginase-driven catabolism.
Taken together, the literature supports several applications of plasma arginine, ornithine, and citrulline measurement in immunometabolism and inflammation:
detecting inflammation-associated arginine deficiency
linking plasma arginine and citrulline to inflammatory severity
monitoring recovery from acute inflammatory states
identifying arginase-driven metabolic rewiring in chronic inflammatory disease
improving interpretation through ratios such as arginine/ornithine and GABR
Applications in endothelial dysfunction and cardiovascular research
Plasma arginine measurement is highly relevant in endothelial and cardiovascular research because arginine is the substrate for nitric oxide synthase, and nitric oxide is a central regulator of endothelial homeostasis, vascular tone, and vascular reactivity. When arginine bioavailability decreases, nitric oxide production may become limited, which is why disturbances in circulating arginine metabolism have long been linked to endothelial dysfunction and cardiometabolic risk.
In this setting, ratios are often more informative than single analytes alone. Absolute plasma arginine can reflect substrate availability, but ratios such as the arginine/ornithine ratio and the global arginine bioavailability ratio (GABR) better capture the balance between arginine availability and its metabolic conversion. This is particularly useful in vascular biology, where the underlying question is often not only how much arginine is present, but also whether it is being preferentially diverted away from nitric oxide production.
A widely cited study by Sourij et al. showed that both GABR and the arginine/ornithine ratio were associated with markers of endothelial dysfunction and with increased cardiovascular mortality risk. These findings support the use of plasma arginine-related ratios as biologically meaningful tools in cardiovascular and cardio-metabolic research, particularly when studying endothelial impairment, vascular risk, or altered nitric oxide-related metabolism.
Applications in intestinal function assessment
Among the three analytes, citrulline has the clearest standalone application in plasma. Because circulating citrulline is produced mainly by small-bowel enterocytes, plasma citrulline is widely recognized as a biomarker of enterocyte functional mass and has been studied in intestinal failure, short bowel syndrome, mucosal injury, and intestinal integrity assessment. Reviews and clinical studies consistently describe reduced plasma citrulline as a marker of impaired intestinal function and reduced absorptive small-bowel mass.
This makes citrulline especially valuable when the main research question is intestinal status rather than broader arginine-pathway balance.
Why the ratios matter
Arginine/Ornithine ratio
Because arginase converts arginine into ornithine, the arginine/ornithine ratio is useful to assess arginase pathway pressure and broader arginine bioavailability. Our Arginine-to-Ornithine ratio pack is positioned specifically for insight into arginine-pathway activity and immune regulation in plasma.
Citrulline/Arginine ratio
The citrulline/arginine ratio is linked to nitric oxide synthase-related biology, since NOS converts arginine into citrulline and nitric oxide. Our Citrulline-to-Arginine ratio ELISA pack is thus indicative of NOS activity in plasma samples.
Global Arginine Bioavailability Ratio (GABR)
Defined as arginine / (ornithine + citrulline), GABR integrates the two main competing fates of arginine and has been proposed as a more sensitive index of arginine bioavailability than arginine concentration alone.
Global Arginine Bioavailability ratio (GABR) ELISA pack
€ 1.499€ 1.887
Immusmol ELISA kits for arginine-pathway monitoring
Immusmol developed a practical set of ELISA tools for arginine-pathway monitoring, designed for easy implementation in research workflows.
Key advantages include low sample volume requirements, cross-species applicability compatible with both human and preclinical studies, and cross-validation of the underlying single-analyte assays against LC-MS.
Together, these assays enable robust measurement of arginine, ornithine, citrulline, and biologically informative related ratios, including in small-animal studies where sample availability may be limited.
| Product | Format | Min. vol. | Validated matrix / positioning |
|---|---|---|---|
| Arginine ELISA kit | Single analyte | 20 µL | Serum, plasma, and cell-culture supernatants; reacts with all species |
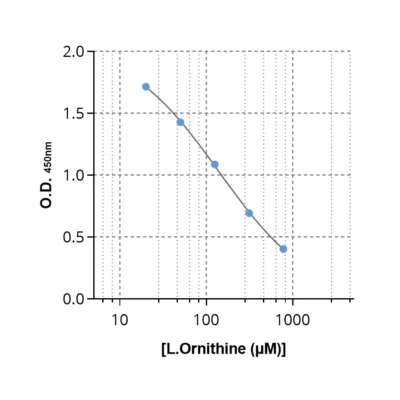
| Ornithine ELISA kit | Single analyte | 20 µL | Plasma; reacts with all species |
| Citrulline ELISA kit | Single analyte | 20 µL | Plasma; reacts with all species |
| Arginine-to-Ornithine Ratio ELISA Pack | Pack | 20 µL | Plasma; reacts with all species |
| Citrulline-to-Arginine Ratio ELISA Pack | Pack | 20 µL | Plasma; reacts with all species |
| GABR ELISA Pack | Pack | 20 µL | Plasma; reacts with all species |
Pre-analytical and assay tips for reliable plasma quantification
To obtain reliable results with arginine-pathway metabolites, careful attention to sample handling is essential. Because arginine, ornithine, and citrulline are linked to active metabolic pathways, pre-analytical variability can affect the measured concentration.
A few simple precautions can help improve data quality and result consistency:
Use EDTA plasma and process blood promptly
For arginine, ornithine, and citrulline, we recommend EDTA plasma and rapid processing after blood collection. This helps limit ex vivo metabolic activity that may alter metabolite levels before analysis.
Keep sample handling consistent throughout the study
Use the same collection tubes, processing conditions, storage workflow, and freeze-thaw policy for all samples. Standardized pre-analytics help reduce variability and make biological differences easier to interpret.
Avoid haemolytic or lipemic samples.
Compromised samples can interfere with assay performance and increase variability. For best robustness, only use clean, well-prepared plasma samples.
Minimize freeze-thaw cycles
Repeated freezing and thawing may affect sample integrity and contribute to variability. When possible, aliquot samples before storage to avoid unnecessary handling.
Work within the validated matrix
Our assays are validated in defined matrices, and performance is best controlled under those conditions. If you plan to use another sample type, we recommend verifying assay performance in your own matrix before starting the study.
Apply careful ELISA technique, especially for ratios
For ratio-based readouts, small variations in either analyte can affect the final result. Running samples in duplicates, using calibrated pipettes, and maintaining consistent incubation and plate-handling conditions are therefore particularly important.
Frequently asked questions
Why measure arginine, ornithine and citrulline together instead of arginine alone?
Because these three metabolites are metabolically interconnected. Measuring arginine alone provides only partial information, whereas adding ornithine and citrulline enables calculation of ratios that help interpret arginase-related metabolism, nitric oxide-associated pathways, and overall arginine bioavailability.
Which ratios are most useful for arginine-pathway interpretation?
The most informative ratios depend on the biological question. Arginine/ornithine is commonly used for arginase-related biology, citrulline/arginine can support interpretation of nitric oxide-related metabolism, and GABR provides an integrated view of arginine bioavailability relative to ornithine and citrulline.
Which analyte is the most useful marker of intestinal function?
Citrulline is the best-established marker among the three. Plasma citrulline is widely described as a marker of enterocyte functional mass and intestinal integrity.
Which sample type is recommended for arginine, ornithine and citrulline measurement?
For the arginine, ornithine and citrulline ELISAs discussed on this page, EDTA plasma is the recommended matrix for routine use. Consistent pre-analytics and prompt sample processing are important to limit ex vivo changes and improve result reliability.
Are ELISA kits a real alternative to LC-MS for arginine-pathway monitoring?
For focused plasma studies, yes. LC-MS remains a powerful reference method, but ELISA kits offer a simpler and more accessible workflow for targeted monitoring of individual metabolites and biologically relevant ratios. This is especially useful when the objective is pathway-focused quantification rather than broad metabolomic profiling.
Which Immusmol format is most useful for pathway interpretation?
If the goal is to understand pathway activity rather than measure a single metabolite, ratio-based packs are often the most informative starting point. Arginine/ornithine is particularly useful for arginase-related interpretation, while citrulline/arginine and GABR can be useful for nitric oxide biology and arginine bioavailability.
Conclusion
Arginine, ornithine, and citrulline should often be interpreted together rather than as isolated metabolites. Because they are metabolically interconnected, their combined measurement — and especially the use of biologically informative ratios such as arginine/ornithine, citrulline/arginine, and GABR — can provide a more useful view of arginase-related activity, nitric oxide-associated metabolism, and overall arginine bioavailability in plasma.
For laboratories seeking a practical alternative to LC-MS for targeted pathway monitoring, ELISA-based workflows offer an accessible solution for focused translational and preclinical studies. With single-analyte kits and ratio-based packs designed for arginine-pathway interpretation, Immusmol helps researchers move from metabolite quantification to clearer biological insight.

Dominique Bodet, PhD
Dominique is R&D Director – Antibodies & Immunoassays at ImmuSmol. A biochemist with 25+ years of experience, he specializes in developing anti-hapten antibodies and immunoassays for the detection and quantification of small molecules. At ImmuSmol, Dominique has supported 600+ customers worldwide in assay selection, validation, and troubleshooting, helping researchers achieve reliable results with antibody- and ELISA-based bioanalytical solutions.
How we wrote this
This guide was written by Immusmol’s scientific team based on (i) recurring questions from researchers monitoring arginine, ornithine, and citrulline in serum and plasma, including interest in their interpretation as single biomarkers or as biologically relevant ratios, (ii) established analytical principles for the quantification of small polar amino-acid metabolites using LC-MS, HPLC, and anti-hapten-based ELISA workflows, and (iii) practical performance and implementation considerations relevant to serum/plasma testing, including pre-analytics, matrix effects, and ratio interpretation. The recommendations are intended to help readers understand the biological relevance of these markers, select an appropriate analytical approach, and implement fit-for-purpose controls to generate robust and reproducible data.
Safety & scope
This article provides technical guidance on the quantification of arginine, ornithine, and citrulline in serum or plasma, with a focus on the biological applications of these analytes and their key ratios, including the arginine/ornithine ratio, the citrulline/arginine ratio, and the global arginine bioavailability ratio (GABR). It discusses the main analytical approaches used to monitor these metabolites, including LC-MS/HPLC-based methods and ELISA workflows, and highlights the main pre-analytical, matrix-related, and assay-implementation factors that can affect data quality. It is intended for research and analytical audiences and focuses on best practices for sample handling, method selection, and basic assay verification. Requirements may vary depending on the study context, whether exploratory, translational, regulated, or clinical; always follow your institution’s SOPs and the instructions for use (IFU) provided with any assay.
Disclaimer
The information provided is for educational and general methodological purposes only. It is not medical advice and must not be used as the sole basis for clinical diagnosis, treatment decisions, or patient management. The biomarkers and ratios discussed in this article may have different levels of validation depending on the intended application, study design, and regulatory setting. Users remain responsible for selecting assays and workflows appropriate to their samples, objectives, and jurisdiction, and for performing any validation or verification required in their laboratory environment.