Comprehensive and easy-to-access evaluation of the Kynurenine pathway by ELISA!
June 23, 2022 2025-06-10 15:05Comprehensive and easy-to-access evaluation of the Kynurenine pathway by ELISA!


Among mechanisms associated with cancer immune evasion, the activation of the Kynurenine Pathway (KP) is known to play a key role and has thus been proposed as a therapeutic target to improve current immune checkpoint blockade-based immunotherapies (Peyraud et al., 2022). The first step of this pathway relies on the activation by inflammatory stimuli (eg. IFNg, IL6, …) of Tryptophan degrading enzymes including IDO1/2 (indoleamine 2,3 dioxygenase) and TDO2 (Tryptophan 2,3 dioxygenase). L-Kynurenine is the first stable metabolite of the KP and can be transformed into a series of metabolites collectively known as Kynurenines and which include among others 3-OH Kynurenine, Kynurenic (KynA), Xanthurenic and Quinolinic (Quin) acids. While the quantification of Kynurenine (Kyn), Tryptophan (Trp) and their related ratio has been widely evaluated in samples from cancer patients, little is known for the other metabolites.
We developed and validated new and easy-to-use immunoassays for the quantification of KynA and Quin, and evaluated their respective abundance in plasma from cancer patients and healthy subjects – KynA to Quin ratio was also calculated. While plasmatic contents of both KynA and Quin were increased in cancer patients compared to healthy subjects, the ratio did not differ thus arguing for the whole KP activation instead of a preferred axis.
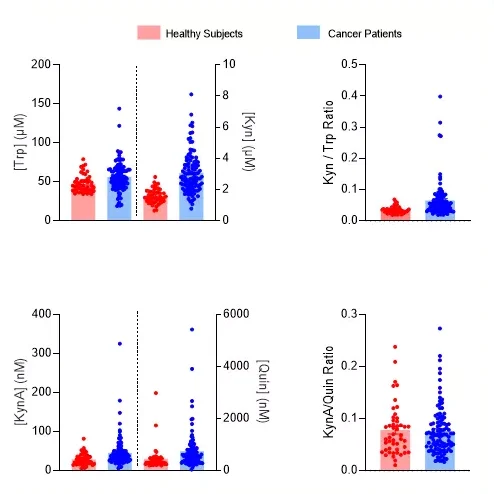
Plasma samples from healthy subjects (n=49) and cancer patients (n=106) were processed for the quantification, through immunoassays, of Kyn (BA-E-2200R), Trp (BA-E-2700R), KynA (IS-I-0200R) and Quin (IS-I-0100R). Each metabolites and ratios – Kyn/Trp & KynA/Quin – are presented as both individual values and means +/- SEM.