Identification of IDO modulators using Kynurenine & Tryptophan as surrogate markers
September 23, 2020 2025-06-10 15:37Identification of IDO modulators using Kynurenine & Tryptophan as surrogate markers

Macrophages represent a wide group of functionally-different subpopulations involved in several immunological functions. In contrast to M1 macrophages which favor a pro-inflammatory response, M2 macrophages trigger inflammation resolution and suppress T cell activation via several mechanisms including release of anti-inflammatory cytokines, immune checkpoint engagement, and metabolic activities such as indoleamine 2,3-dioxygenase (IDO), which concomitantly promotes Tryptophan degradation and Kynurenines production.
Novel therapeutic options in immuno-oncology consist in manipulating – directly or indirectly – such a metabolic pathway to relieve the immunosupressive functions of M2 macrophages. Indeed, IDO can be modulated directly through conventional pharmacological inhibition (eg. Epacadostat) or indirectly through modulation of specific signaling pathways (eg. PI3K). As to investigate innovative direct or indirect modulators of IDO activity, Kynurenine and Tryptophan represent valuable biomarkers to be measured and can easily be adressed through ELISA kits which thus offer screening capacities.
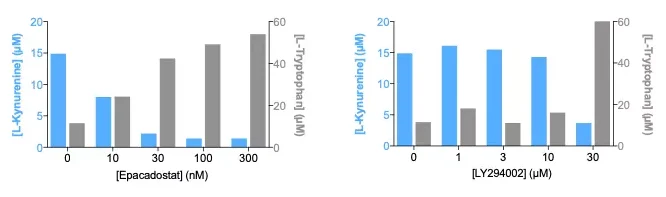
IDO activity of M2 polarized macrophages is decreased by its pharmacological blockade but also through PI3K signaling pathway inhibition. M2-macrophages were treated during the polarization with increasing concentrations of Epacadostat – as a prototypical IDO inhibitor – or LY294002 – a reference PI3K inhibitor. ELISA-based quantification of L-Tryptophan and L-Kynurenine levels in the supernatants (using BA-E-2700 and BA-E-2200 kits, respectively) revealed a dose-dependent inhibition of activated IDO as highlighted by the increase and decrease of Tryptophan and Kynurenine, respectively.