Assessing the polyamine pathway through immunoassays!
September 7, 2023 2025-06-05 16:33Assessing the polyamine pathway through immunoassays!

Assessing the polyamine pathway through immunoassays!
Scientific background
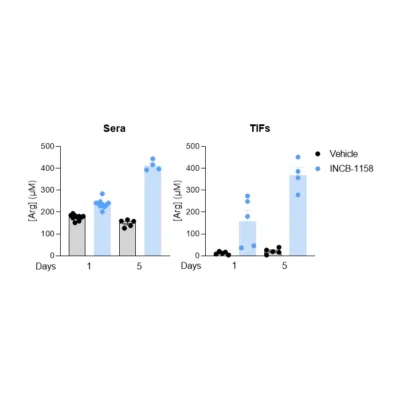
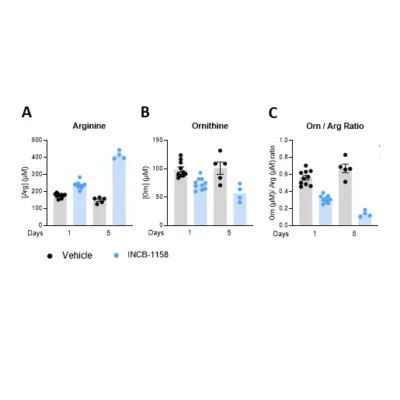
Polyamines are molecules derived from ornithine (Orn) (i.e., putrescine, spermine, spermidine) or arginine (Arg) (i.e., agmatine). They are present in mammalian cells at millimolar concentrations1. Key regulatory steps of their metabolism are represented by ornithine decarboxylase (ODC), acetyl-CoA:spermidine/spermine N1-acetyltransferase (cSAT), acetylCoA:spermidine-N8-acetyltransferase (nSAT), and membrane transport systems. While ODC is responsible for ornithine degradation and spermidine production, cSAT and nSAT transform spermidine to N1- and N8-acetylspermidine (N1- and N8-AcSpdn), respectively. These molecules are involved in many critical processes such as cell proliferation, nucleic acid synthesis, and cytoprotection from oxidative stress2. Several studies have evidenced that polyamine metabolism is frequently increased in cancer3, which can fuel tumor progression4 and favor immune escape5. Elevated levels of polyamines in sera of cancer patients have been demonstrated to correlate with poor prognosis6.
Our new ELISA kits
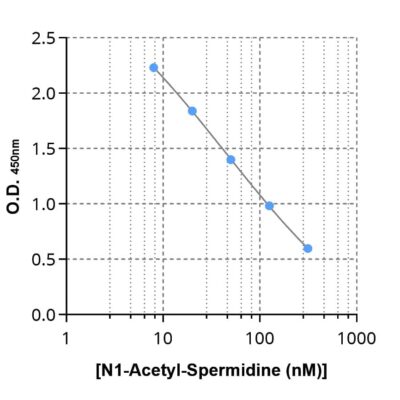
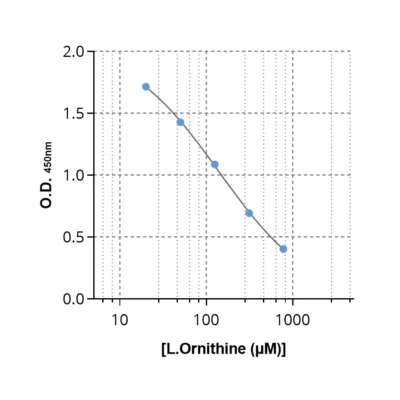
Given the importance of this metabolic pathway in cancer progression4 and resistance to treatment7, investigating it requires robust tools. To this aim, we developed and validated the first ELISA kits to quantify ornithine (#IS-I-1000R), N1-acetylspermidine (#IS-I-2400R), and N8-acetylspermidine (#IS-I-2500R) in samples of different natures (plasma, cell culture supernatants) and species (mouse, human).
Illustrative data
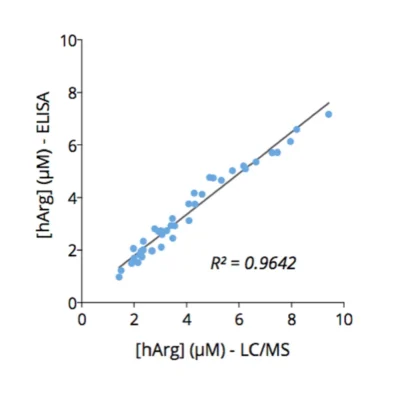
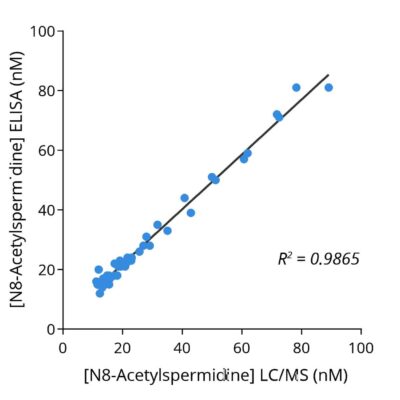
Our immunoassay specifications were first determined by comparing quantified analyte levels, in the same plasma samples, using conventional mass spectrometry and our ELISA kits (Figure 1), thus validating the technological platform. Finally, using these immunoassays, we evaluated the levels of the three analytes in cell culture supernatants from MC38 (mouse, colon), 4T1 (mouse, breast), HCT116 (human, colon) and BT549 (human, breast) tumor cell lines cultured over a 48-hour period in the presence and absence of the prototypical ODC inhibitor, D,L-alpha-difluoromethylornithine (DFMO). As depicted in Figure 2, Orn is differentially consumed in the various cell lines, with a maximum observed in HCT116 cells. Concomitantly, while N1-AcSpdn is detected at different levels in all tumor cell lines, N8-AcSpdn is produced at significant amount only in HCT116 cells, thus arguing for a differential activation level of the polyamine pathway. This dataset confirms the robustness of our ELISA kits to address the polyamine pathway.
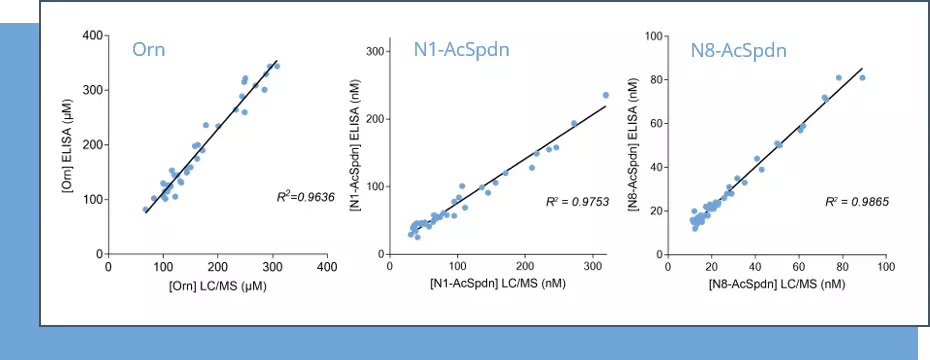
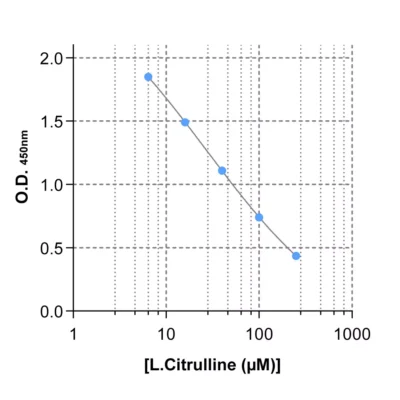
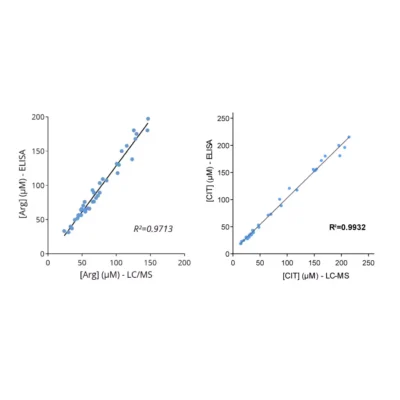
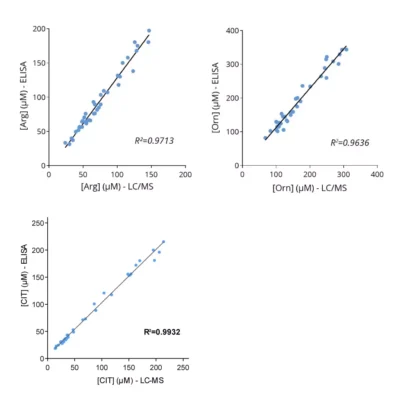
Figure 1 – Cross-validation of ELISA and LC/MS data in human serum samples. Human serum samples (n=40) were processed for Orn, N1- and N8-AcSpdn measurements by mean of both ELISA kits and LC/MS. As depicted, the results obtained with ELISA and LC/MS are highly correlated (R Squared > 0,95).
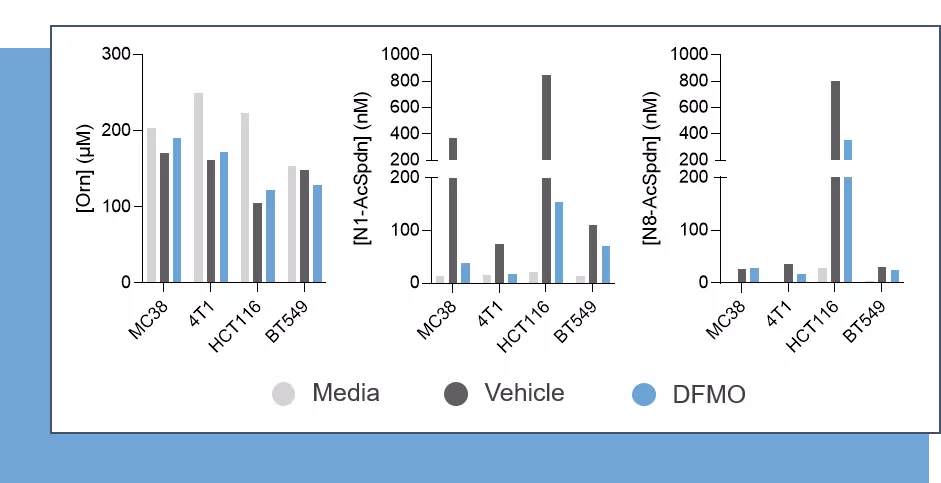
Figure 2 – Cancer cell lines harbor a differential polyamine pathway activity, which is limited upon ODC inhibition. Mouse (MC38 & 4T1) and human (HCT116 & BT549) tumor cell lines were seeded for a 24 hour period and then exposed or not to DFMO – an ODC inhibitor – for two days before supernatant collection and assessment of Orn, N1- and N8-AcSpdn levels using ELISA kits (IS-I-1000R, IS-I-2400R and IS-I-2500R, respectively).
Our ELISA kits to measure arginine metabolites:
Global Arginine Bioavailability ratio (GABR) ELISA pack
€ 1.499€ 1.887
N1-Acetyl-Spermidine ELISA kit
€ 629
N8-Acetyl-Spermidine ELISA kit
€ 629