Quantification of L-Arginine in various samples made simple!
December 1, 2022 2025-06-05 16:57Quantification of L-Arginine in various samples made simple!
Quantification of L-Arginine in various samples made simple!
Immunometabolism is known to play a key role in shaping the immune system. Amino acid degradation by tumor and/or suppressive immune cell subsets has been shown to limit anti-tumor immune response (1) and, in this era, L-Arginine (Arg) figures as one of the keystones (2). Indeed, Arg can be degraded within the tumor microenvironment (TME) via Arginase (types 1 & 2) expressed by tumor cells or immune cell subsets including Myeloid Derived Suppressive Cells (MDSCs), which catabolize it into Ornithine plus Urea. Thus, blocking Arginase represents an attractive therapeutic strategy to restore Arg level within the TME and favor the initiation of an effective anti-tumor immune response (3).
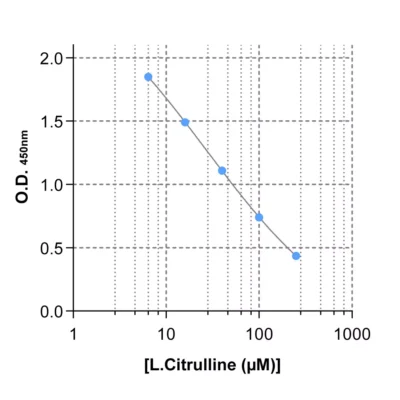
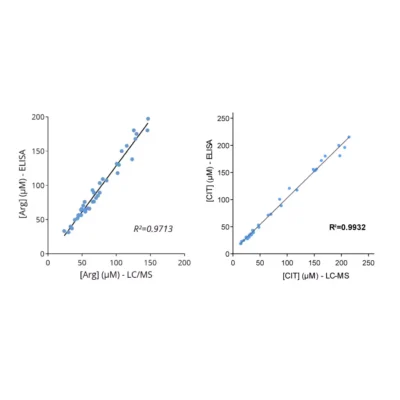
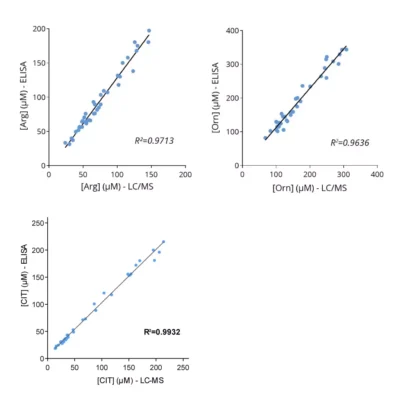
Given the importance of this amino acid in immune regulation, we developed and validated a robust and easy-to-use immunoassay (#IS-I-0400R) for the detection and quantification of Arg in several biological sample types, eg. plasma, cell culture supernatants and tumor interstitial fluid (TIF).
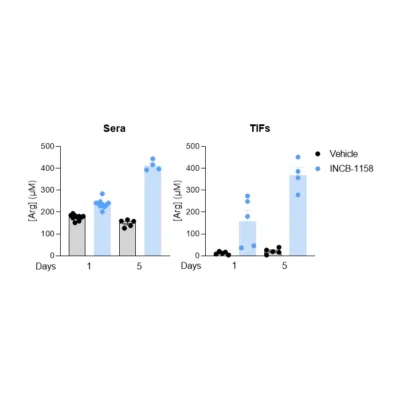
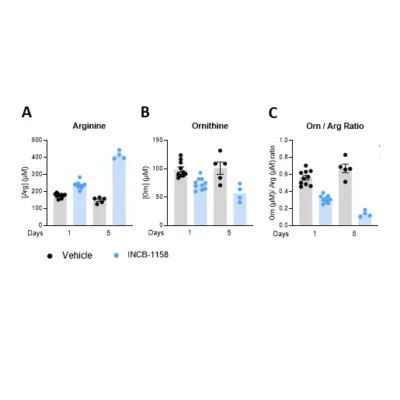
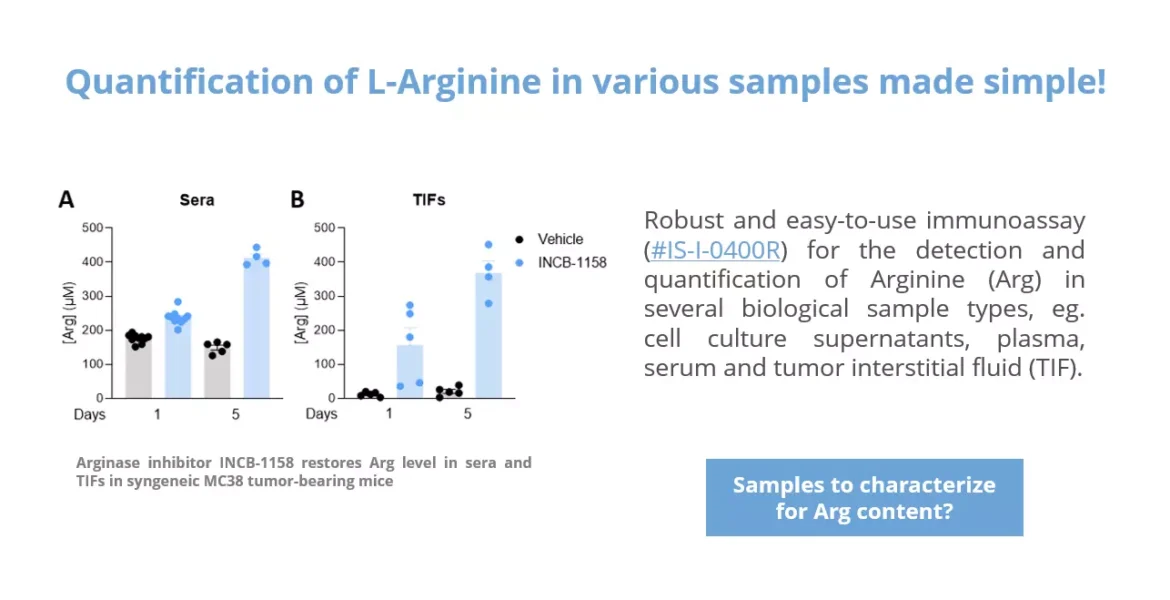
Using a syngeneic MC38 mouse colorectal tumor model along with our immunoassay, we evaluated Arg level within sera and TIFs upon animal treatment with the prototypical Arginase inhibitor, ie. INCB-1158, or with its respective vehicle solution for one or five days. In line with the literature (4), this assay revealed the ability of INCB-1158 to restore Arg level in the periphery (sera) and within the tumor (TIFs), an effect more visible with a longer treatment period. This dataset confirms the robustness of our ELISA kit to quantify Arg in complex biological samples and enlighten Arg metabolism-modulating effects.
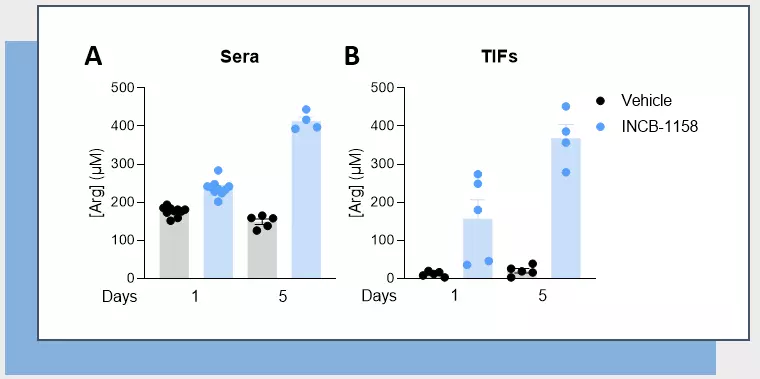
Figure 1 – Arginase inhibitor INCB-1158 restores Arg level in sera and TIFs in syngeneic MC38 tumor-bearing mice. C57BL/6J mice were subcutaneously inoculated with MC38 colorectal tumor cells and treated with INCB-1158 for one or five days. Sera (A) and TIFs (B) were then collected and processed for Arg level quantification using #IS-I-0400R Arg ELISA kit.
Our novel competitive ELISA kit has already been highlighted in a recent study supervised by Antoine Italiano and conducted in collaboration between teams from Institut Bergonié, Institut Gustave Roussy, and Explicyte and published in Annals of Oncology (5). In this study, Peyraud et al. indeed demonstrated that low plasmatic level of Arg was associated with a worse clinical outcome in advanced cancer patients treated with Immune Checkpoint Blockers (ICB), as illustrated through response rate (A) and progression free survival (B).
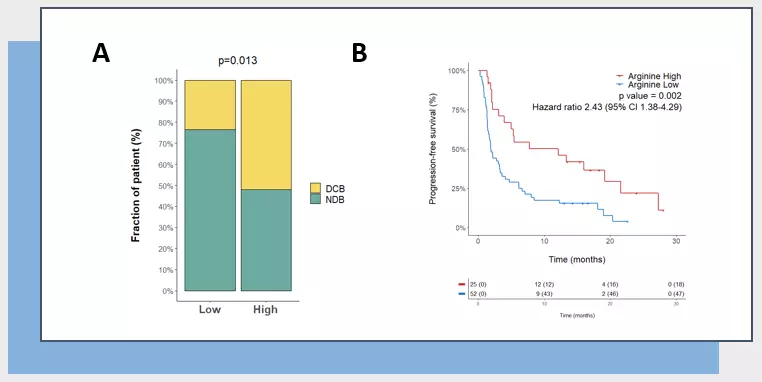
Figure 2 – Arg metabolism is predictive of anti-PD(L)1 immunotherapy-treated patient outcome. (A) Rate of clinical benefit (NDB; Non-durable vs DCB, durable clinical benefit) of advanced cancer patients treated with ICB according to baseline plasmatic Arg level (Low vs High). p-value is calculated using Khi-deux test. (B) Kaplan Meier curves of progression-free survival according to baseline plasmatic Arg levels.
Our ELISA kits to assess arginine metabolism:
Global Arginine Bioavailability ratio (GABR) ELISA pack
€ 1.499€ 1.887
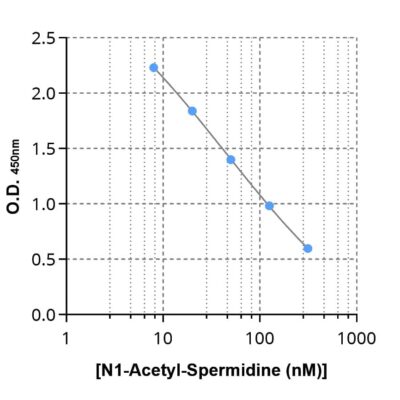
N1-Acetyl-Spermidine ELISA kit
€ 629
N8-Acetyl-Spermidine ELISA kit
€ 629