New L-Glutamine ELISA as tool to address metabolic features in cell culture supernatants
May 19, 2021 2025-06-10 15:28New L-Glutamine ELISA as tool to address metabolic features in cell culture supernatants
New L-Glutamine ELISA as tool to address metabolic features in cell culture supernatants
L-Glutamine (Gln) – a conditionally essential amino acid – is the most abundant free amino acid in human blood. As a source of carbon and nitrogen, Gln supplies many intermediates required for multiple metabolic functions including, among others, purine, pyrimidine, and glutathione synthesis pathways. In addition, one of the important functions of Gln – once deaminated into Glutamate by Glutaminase (Gls1 & Gls2) – is to fuel the citric acid cycle (TCA), which represents a major energy-producing process, especially for highly proliferating cells e.g. T lymphocytes and cancer cells. Hence, because of the cohabitation of both cell types within tumors and the high rate of tumoral glutaminolysis, immune cells are likely to be often deprived of Gln, a condition which can severely limit their functions.
Given its pleiotropic role, targeting Gln metabolism thus represents an attractive druggable therapeutic avenue in immuno-oncology. For instance, Gls1 can be directly targeted through Glutaminase inhibitors such as CB-839 (under clinical development in various type of cancers). Overall, assessing novel therapeutic molecules aiming at restoring Gln level should rely, mainly, on Gln quantification, which can now be addressed by ELISA!
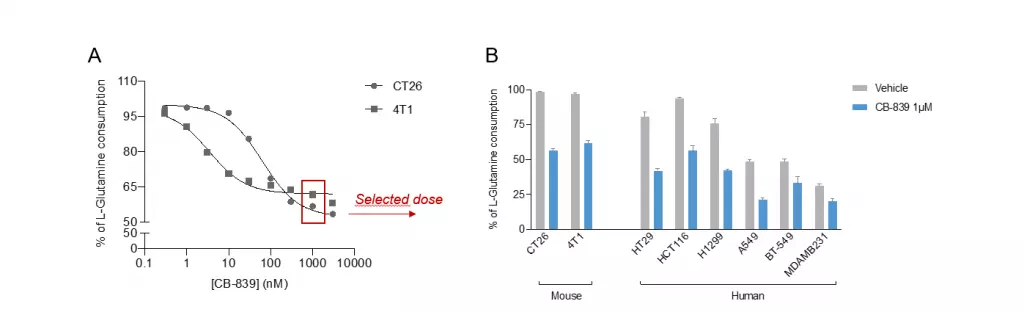
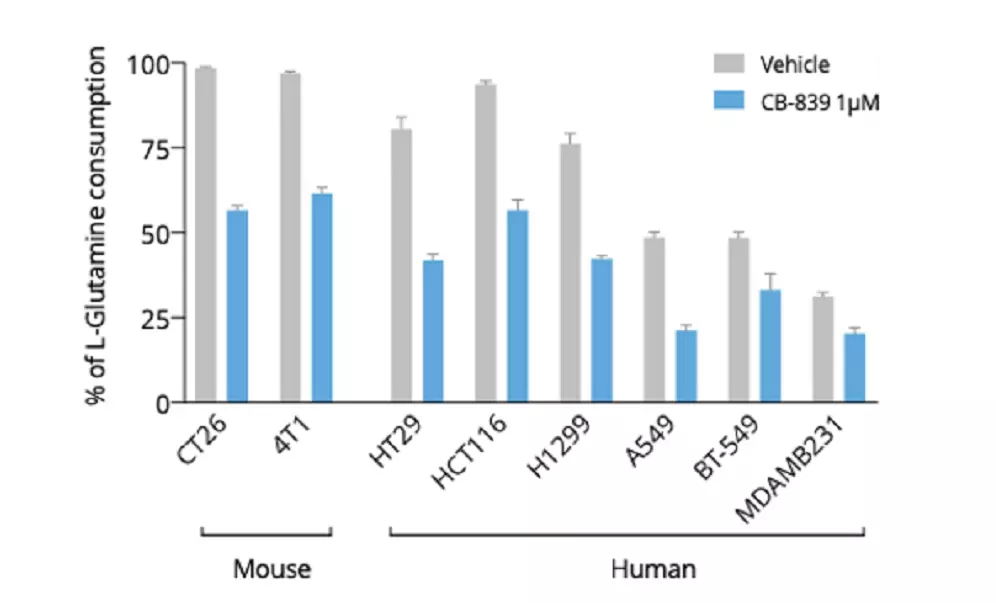
Tumor cell lines from both murine and human origins degrade Glutamine to support their high proliferating rate in a Gls1 dependent manner. A. Mouse 4T1 breast and CT26 colorectal cancer cell lines were treated with increasing concentrations of Gls1 inhibitor (CB-839) for 48 hours and at the end of the treatment period, cell culture supernatants were collected for Glutamine quantification using our ELISA kit (#IS-I-1100). As shown here, both cell lines harbor a high Glutamine consumption rate which is dose dependently reverted by Gls1 blockade. Also, it’s noteworthy that Gls1 blockade by CB-839 only partially reversed Glutamine consumption, thus indicating the participation of another catabolic pathway, more presumably Gls2. B. Using the same experimental design, both mouse and human cancer cell lines were treated with 1µM CB-839, and Glutamine content was evaluated at the end of the treatment using our ELISA kit (#IS-I-1100). As depicted here, the different tumor cell lines harbored a variable Glutamine consumption rate – being higher in murine than in human tumor cell lines. Among the different human cell lines, a higher rate of Glutamine degradation is observed for colorectal (HT29 & HCT116) followed by lung (H1299 & A549) and breast (BT-549 & MDA-MB231) cancer cell lines.
